Embolization Particles Market Size, Share and Trends 2026 to 2035
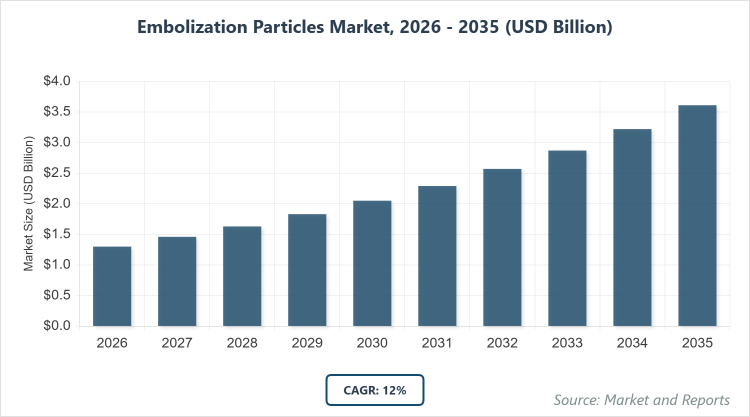
According to MarketnReports, the global Embolization Particles market size was estimated at USD 1.3 billion in 2025 and is expected to reach USD 4 billion by 2035, growing at a CAGR of 12% from 2026 to 2035. Embolization Particles Market is driven by the rising prevalence of cancer and minimally invasive procedures.
What is Embolization Particles? An Industry Overview
Embolization particles are microscopic spheres or agents used in interventional radiology to block blood vessels, treating conditions like tumors, aneurysms, or hemorrhages by restricting blood flow to targeted areas, minimizing surgery risks and promoting patient recovery. The market definition includes biocompatible materials such as microspheres, drug-eluting beads, and PVA particles designed for precise delivery via catheters, supporting therapeutic applications in oncology, vascular medicine, and urology while adhering to regulatory standards for safety and efficacy to advance minimally invasive treatments and improve clinical outcomes in healthcare.
What are the Key Insights of Embolization Particles Market?
- The global Embolization Particles market was valued at USD 1.3 billion in 2025 and is projected to reach USD 4 billion by 2035.
- The global Embolization Particles market is expected to grow at a CAGR of 12% during the forecast period 2026-2035.
- The Embolization Particles market is driven by increasing cancer incidences and advancements in interventional radiology techniques.
- The microspheres subsegment dominates the type segment with a 40% share due to their uniform size and controlled release properties for targeted therapy.
- The tumor treatment subsegment dominates the application segment with a 45% share owing to rising oncology cases requiring embolization for tumor embolization.
- The hospitals subsegment dominates the end-user segment with a 60% share because of advanced infrastructure for interventional procedures.
- North America dominates the global Embolization Particles market with a 35% share attributed to high healthcare spending and technological innovations in the U.S.
What are the Market Dynamics of Embolization Particles?
Growth Drivers
The embolization particles market is propelled by the growing prevalence of cancer worldwide, with over 19 million new cases annually as reported by WHO, driving demand for minimally invasive treatments like transarterial chemoembolization (TACE) that utilize drug-eluting beads for targeted drug delivery, reducing side effects and improving patient outcomes. Technological advancements in particle design, such as bioresorbable materials that degrade after treatment, enhance safety and efficacy, attracting investments from medical device firms. Regulatory approvals, like FDA’s fast-track for innovative particles, accelerate adoption, while aging populations in developed regions increase procedures for vascular conditions, fostering market expansion through hospital procurements.
Restraints
High costs of embolization procedures, often exceeding USD 10,000 per session due to specialized particles and imaging equipment, limit accessibility in developing regions with constrained healthcare budgets. Stringent regulatory requirements for biocompatibility and clinical trials, averaging 3-5 years for approval, delay product launches and increase R&D expenses. Additionally, risks of complications like non-target embolization require skilled interventional radiologists, whose shortage in rural areas hinders market penetration and adoption in underserved markets.
Opportunities
The integration of embolization particles with imaging technologies like MRI-compatible beads offers opportunities for precise navigation, potentially reducing procedure times by 20% and expanding applications in neurology for aneurysm treatment. Emerging markets in Asia-Pacific present growth through rising healthcare infrastructure, where affordable PVA particles can address high liver cancer rates. Partnerships for drug-loaded particles in oncology, combining with immunotherapies, can unlock new therapeutic avenues, capitalizing on the global oncology market’s expansion.
Challenges
Ensuring particle uniformity and controlled degradation poses manufacturing challenges, as variations can lead to inconsistent therapeutic outcomes, requiring advanced quality control that elevates costs. Reimbursement inconsistencies across insurers and regions limit procedure volumes, while patient awareness gaps in non-oncology applications hinder demand. Environmental concerns over non-biodegradable particles demand sustainable alternatives, adding R&D complexity amid supply chain issues for raw materials.
Embolization Particles Market: Report Scope
| Report Attributes | Report Details |
| Report Name | Embolization Particles Market |
| Market Size 2025 | USD 1.3 Billion |
| Market Forecast 2035 | USD 4 Billion |
| Growth Rate | CAGR of 12% |
| Report Pages | 220 |
| Key Companies Covered | Boston Scientific, Merit Medical, Sirtex Medical, Terumo Corporation, BTG (Varian), Cook Medical, and Others |
| Segments Covered | By Type, By Application, By End-User, and By Region |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, and The Middle East and Africa (MEA) |
| Base Year | 2025 |
| Historical Year | 2020 – 2024 |
| Forecast Year | 2026 – 2035 |
| Customization Scope | Avail customized purchase options to meet your exact research needs. |
The Embolization Particles market is segmented by type, application, end-user, and region.
Based on Type Segment: The microspheres segment is the most dominant, holding a 40% market share, as they provide precise sizing for vascular occlusion, driven by their use in radioembolization for liver tumors. This dominance drives market growth by enabling targeted therapies with minimal side effects. The drug eluting beads (DEBs) segment is the second most dominant with a 30% share, allowing controlled drug release, which propels the market through combination therapies in oncology.
Based on Application Segment: The tumor treatment segment is the most dominant, capturing a 45% share, due to the high incidence of cancers requiring embolization. Its dominance accelerates market growth by supporting minimally invasive alternatives to surgery. The uterine fibroid embolization (UFE) segment ranks second with a 25% share, addressing women’s health issues, driving the market with outpatient procedures.
Based on End-User Segment: The hospitals segment is the most dominant, with a 60% share, attributed to advanced interventional suites. This leads market growth by facilitating complex procedures. The specialty clinics segment is second with a 25% share, offering focused care, propelling growth through ambulatory settings.
What are the Recent Developments in Embolization Particles Market?
- In 2025, Boston Scientific launched bioresorbable microspheres for temporary embolization, improving patient recovery times.
- Merit Medical expanded its PVA particle line with enhanced sizing for precision in UFE procedures.
- Sirtex Medical partnered with a pharma firm for next-gen radioembolization spheres targeting liver cancer.
- Terumo Corporation introduced drug-eluting beads with extended release for tumor therapy.
- BTG (Varian) acquired a startup for gelatin sponge particles, focusing on hemorrhage control.
What is the Regional Analysis of Embolization Particles Market?
- North America to dominate the global market
North America holds a 35% share in the Embolization Particles market, driven by advanced healthcare and high cancer prevalence. The United States dominates, with Boston Scientific and Merit Medical leading innovations, supported by FDA approvals.
Europe maintains a strong position in the Embolization Particles market, fueled by research funding. Germany leads, with Sirtex and Terumo advancing in oncology, aligned with EU health directives.
Asia Pacific is the fastest-growing region for Embolization Particles, propelled by rising procedures. China dominates, with local firms scaling for liver cancer treatments amid healthcare expansions.
Latin America is emerging in the Embolization Particles market, focusing on vascular care. Brazil leads, adopting for hemorrhage control in public hospitals.
The Middle East and Africa show potential in Embolization Particles, driven by medical tourism. The UAE is a key player, importing for specialty clinics in Dubai.
Who are the Key Market Players and Strategies in Embolization Particles?
Boston Scientific focuses on bioresorbable innovations, launching for improved recovery.
Merit Medical emphasizes PVA expansions, targeting precision procedures.
Sirtex Medical pursues partnerships for radioembolization advancements.
Terumo Corporation introduces extended-release DEBs, focusing on tumors.
BTG (Varian) acquires for gelatin particles, addressing hemorrhage.
Cook Medical develops microspheres for vascular applications, expanding portfolios.
What are the Market Trends in Embolization Particles?
- Increasing adoption of bioresorbable particles for temporary occlusion.
- Growth in drug-eluting beads for targeted chemotherapy.
- Focus on precision sizing for minimally invasive procedures.
- Expansion of radioembolization for liver cancer treatments.
- Shift toward PVA and gelatin for cost-effective options.
- Integration with imaging for real-time guidance.
What are the Market Segments and their Subsegment Covered in the Embolization Particles Report?
By Type
-
- Radioembolization Spheres
- Microspheres
- Drug Eluting Beads (DEBs)
- PVA Particles
- Gelatin Sponge Particles
- Others
By Application
-
- Uterine Fibroid Embolization (UFE)
- Tumor Treatment
- Hemorrhage Control
- Liver Cancer Embolization
- Prostate Artery Embolization
- Varicocele Embolization
- Others
By End-User
-
- Hospitals
- Specialty Clinics
- Ambulatory Surgical Centers
- Diagnostic Centers
- Research Institutes
- Others
By Region
-
- North America
- U.S.
- Canada
- Europe
- UK
- Germany
- France
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Rest of Asia Pacific
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Middle East & Africa
- UAE
- South Africa
- Rest of Middle East & Africa
- North America
Chapter 1. Preface
Chapter 2. Executive Summary
Chapter 3. Global Embolization Particles Market - Industry Analysis
Chapter 4. Global Embolization Particles Market- Competitive Landscape
Chapter 5. Global Embolization Particles Market - Type Analysis
Chapter 6. Global Embolization Particles Market - Application Analysis
Chapter 7. Global Embolization Particles Market - End-User Analysis
Chapter 8. Embolization Particles Market - Regional Analysis
Chapter 9. Company Profiles
Frequently Asked Questions
Embolization Particles are microscopic agents used to block blood vessels in medical procedures for treating tumors or hemorrhages.
Key factors include cancer prevalence, minimally invasive procedure demand, and technological advancements.
The market is projected to grow from USD 1.4 billion in 2026 to USD 4 billion by 2035.
The market is expected to grow at a CAGR of 12% during 2026-2035.
North America will contribute notably, holding a 35% share due to advanced healthcare.
Major players include Boston Scientific, Merit Medical, Sirtex Medical, Terumo Corporation, BTG (Varian), and Cook Medical.
The report provides market size, trends, segmentation, regional insights, key players, and forecasts.
Stages include raw material sourcing, manufacturing, testing, distribution, and clinical application.
Trends favor bioresorbable and drug-eluting particles, with preferences for minimally invasive options.
FDA approvals and biocompatibility standards drive innovation in safe materials.