Blood Clot Retrieval Devices Market Size, Share and Trends 2026 to 2035
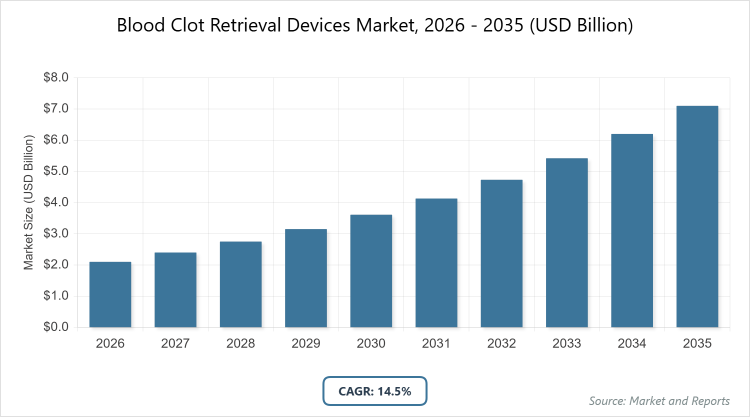
According to MarketnReports, the global Blood Clot Retrieval Devices Market size was estimated at USD 2.1 billion in 2025 and is expected to reach USD 7.1 billion by 2035, growing at a CAGR of 14.5% from 2026 to 2035. Blood Clot Retrieval Devices Market is driven by the rising incidence of ischemic strokes and technological advancements in minimally invasive thrombectomy procedures.What are the Key Insights into the Blood Clot Retrieval Devices Market?
- The global blood clot retrieval devices market was valued at USD 2.1 billion in 2025 and is projected to reach USD 7.1 billion by 2035.
- The market is expected to grow at a CAGR of 14.5% from 2026 to 2035.
- The market is driven by increasing prevalence of ischemic strokes, growing adoption of minimally invasive procedures, and advancements in device technology.
- Mechanical embolus removal devices dominate the device type segment with approximately 45% share due to their high efficacy in large vessel occlusions and proven clinical success rates.
- Ischemic stroke leads the stroke type segment with around 70% share owing to its high incidence and the critical need for rapid reperfusion therapies.
- Hospitals dominate the end-user segment with about 72% share attributed to their role as primary centers for emergency interventions and advanced surgical capabilities.
- North America holds the largest regional share at about 39% thanks to advanced healthcare infrastructure, high stroke prevalence, and favorable reimbursement policies.
What is the Industry Overview of the Blood Clot Retrieval Devices Market?
The blood clot retrieval devices market comprises medical instruments designed to remove thrombi or emboli from blood vessels, primarily to restore blood flow in cases of acute blockages. Market definition refers to the global sector focused on the innovation, manufacturing, distribution, and clinical application of these devices, which include stent retrievers, aspiration systems, and mechanical thrombectomy tools used in endovascular procedures. This market supports interventional radiology and neurology by enabling rapid, minimally invasive interventions that reduce mortality and disability from conditions like stroke, pulmonary embolism, and peripheral artery disease, while adapting to advancements in imaging guidance and biocompatible materials to improve procedural outcomes and patient recovery times.
What are the Market Dynamics of the Blood Clot Retrieval Devices Market?
Growth Drivers
The expansion of the blood clot retrieval devices market is fueled by the surging global incidence of cardiovascular and cerebrovascular diseases, particularly ischemic strokes, which demand urgent and effective clot removal to prevent long-term neurological damage. Technological innovations, such as next-generation stent retrievers with enhanced navigability and aspiration systems integrated with real-time imaging, have improved procedural success rates and reduced complication risks, encouraging wider adoption among interventional specialists. Rising awareness campaigns by health organizations, coupled with aging populations in developed nations and increasing healthcare investments in emerging markets, further accelerate demand. Additionally, supportive regulatory approvals for extended treatment windows and combination therapies with thrombolytics contribute to market growth by expanding patient eligibility and enhancing overall therapeutic outcomes.
Restraints
Market progression is hindered by the high costs of advanced retrieval devices and associated procedures, which restrict accessibility in low-resource settings and strain healthcare budgets in developing regions. Limited availability of skilled neurointerventionalists and specialized facilities poses challenges, particularly in rural areas, leading to delays in treatment and suboptimal utilization. Stringent regulatory requirements for device approvals, including extensive clinical trials to demonstrate safety and efficacy, can prolong time-to-market and increase development expenses. Moreover, concerns over potential complications like vessel perforation or distal embolization, along with variability in reimbursement policies across countries, deter widespread adoption and impact market penetration.
Opportunities
Emerging prospects lie in the development of AI-assisted devices that offer predictive analytics for clot composition and optimal retrieval strategies, potentially revolutionizing procedural precision and personalization. Untapped markets in Asia-Pacific and Latin America offer growth through rising healthcare infrastructure investments and increasing stroke awareness programs, creating demand for affordable, user-friendly devices. Partnerships between device manufacturers and telemedicine platforms can extend reach to remote areas, enabling faster diagnosis and intervention. Furthermore, ongoing research into bioresorbable materials and hybrid devices combining aspiration and stenting could address current limitations, opening avenues for new product launches and market differentiation.
Challenges
Navigating diverse regulatory environments across regions presents hurdles, as differing standards for clinical evidence and post-market surveillance can complicate global expansion and increase compliance costs. Intense competition among established players fosters price pressures and demands continuous innovation, challenging smaller firms to sustain R&D investments. Supply chain disruptions for specialized components, such as nitinol alloys, exacerbate manufacturing delays and cost fluctuations. Additionally, the need for comprehensive training programs to ensure procedural proficiency among healthcare providers remains a barrier, as inadequate expertise can lead to underutilization and affect market trust.
Blood Clot Retrieval Devices Market: Report Scope
| Report Attributes | Report Details |
| Report Name | Blood Clot Retrieval Devices Market |
| Market Size 2025 | USD 2.1 Billion |
| Market Forecast 2035 | USD 7.1 Billion |
| Growth Rate | CAGR of 14.5% |
| Report Pages | 220 |
| Key Companies Covered |
Medtronic plc, Stryker Corporation, Boston Scientific Corporation, Penumbra, Inc., Johnson & Johnson (DePuy Synthes), and Others. |
| Segments Covered | By Device Type, By Stroke Type, By Application, By End-User, and By Region. |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, and The Middle East and Africa (MEA) |
| Base Year | 2025 |
| Historical Year | 2020 - 2024 |
| Forecast Year | 2026 - 2035 |
| Customization Scope | Avail customized purchase options to meet your exact research needs. |
How is the Market Segmentation of the Blood Clot Retrieval Devices Market?
The Blood Clot Retrieval Devices Market is segmented by device type, stroke type, application, end-user, and region.
By Device Type Segment: The mechanical embolus removal devices segment is the most dominant, holding approximately 45% market share, followed by stent retrievers as the second most dominant. Mechanical embolus removal devices dominate due to their versatility in handling various clot types and sizes, particularly in acute ischemic strokes, which drives the market by enabling faster recanalization and better patient outcomes; stent retrievers follow closely, excelling in large vessel occlusions with high retrieval rates, contributing to overall growth through improved procedural efficiency and reduced disability rates.
By Type Segment: Ischemic stroke applications lead as the most dominant segment with around 70% share, with transient ischemic attack as the second most dominant. Ischemic stroke's dominance arises from its prevalence as the primary cause of stroke-related morbidity, necessitating immediate clot retrieval to restore cerebral blood flow, thereby propelling market growth via increased procedure volumes; transient ischemic attacks support this by serving as warning signs that prompt preventive interventions, enhancing demand for devices in early-stage management.
By Application Segment: Neurovascular applications represent the most dominant with about 60% share, while cardiovascular is the second most dominant. Neurovascular dominates owing to the critical time-sensitive nature of brain clot removal in preventing irreversible damage, fueling market expansion through specialized neurointerventional advancements; cardiovascular applications contribute by addressing coronary and cardiac thrombi, broadening device utility and supporting growth in integrated vascular care.
By End-User Segment: Hospitals are the most dominant end-user with around 72% share, whereas ambulatory surgical centers are the second most dominant. Hospitals lead due to their equipped emergency departments and multidisciplinary teams handling high-acuity cases, driving market growth through centralized high-volume procedures; ambulatory surgical centers aid by offering cost-effective outpatient options for less complex cases, increasing accessibility and procedural throughput.
What are the Recent Developments in the Blood Clot Retrieval Devices Market?
- In June 2025, researchers introduced an advanced AI-integrated clot retrieval system designed to improve real-time surgical guidance and enhance precision during stroke procedures.
- In January 2023, Imperative Care, Inc. received U.S. FDA approval for its Zoom RDL Radial Access System, developed specifically to treat ischemic stroke with improved access and control.
- In September 2021, Abbott acquired Walk Vascular, LLC, to expand its peripheral vascular portfolio with minimally invasive thrombectomy systems.
- In October 2025, Stryker announced the launch of an enhanced Trevo XP ProVue Retriever, featuring improved visibility and clot integration for better outcomes in large vessel occlusions.
- In August 2022, phenox GmbH introduced its pRESET mechanical thrombectomy device in Brazil, expanding geographical presence for acute ischemic stroke treatment.
How is the Regional Analysis of the Blood Clot Retrieval Devices Market?
North America to dominate the global market.
North America commands the blood clot retrieval devices market, bolstered by cutting-edge medical technologies, high stroke incidence rates, and robust reimbursement frameworks; the United States dominates within the region with over 85% share, driven by extensive neurointerventional centers, FDA approvals for innovative devices, and national initiatives like the American Heart Association's stroke awareness campaigns that promote timely interventions.
Europe maintains a strong position, characterized by collaborative research networks and standardized treatment protocols across the EU; Germany leads the region, supported by its advanced manufacturing base for medical devices and government-funded programs for stroke prevention, with the UK contributing through NHS investments in thrombectomy services to reduce stroke mortality.
Asia-Pacific demonstrates rapid growth, fueled by urbanization, lifestyle changes increasing stroke risks, and expanding healthcare access; China dominates, leveraging its massive population and state-backed healthcare reforms to boost device adoption, while India advances through affordable innovations and public-private partnerships enhancing rural stroke care infrastructure.
Latin America experiences steady development, propelled by improving economic conditions and rising medical tourism; Brazil holds the lead, facilitated by universal health coverage under SUS and increasing installations of cath labs for endovascular procedures, with Mexico gaining from cross-border collaborations with U.S. firms for technology transfer.
The Middle East and Africa offer nascent potential, constrained by infrastructural gaps but progressing via foreign investments; the UAE and South Africa dominate, with the UAE utilizing oil revenues for high-end hospitals equipped with latest devices, and South Africa benefiting from academic research hubs focusing on vascular health in diverse populations.
Who are the Key Market Players in the Blood Clot Retrieval Devices Market?
Medtronic plc. Medtronic plc adopts strategies centered on product innovation and acquisitions, such as expanding its Solitaire thrombectomy portfolio, to solidify leadership in neurovascular interventions and capitalize on growing demand for ischemic stroke treatments.
Stryker Corporation. Stryker Corporation focuses on R&D investments and clinical collaborations, exemplified by enhancements to its Trevo retriever line, aiming to improve procedural efficacy and expand market share in global thrombectomy applications.
Boston Scientific Corporation. Boston Scientific Corporation pursues geographic expansions and technology integrations, like advancing its Embold detachment systems, to address diverse vascular needs and drive growth through specialized cardiovascular solutions.
Penumbra, Inc. Penumbra, Inc. emphasizes AI-driven platforms and system optimizations, such as the Lightning aspiration technology, to enhance real-time clot removal and target emerging markets for sustained competitive edge.
Johnson & Johnson (DePuy Synthes). Johnson & Johnson (DePuy Synthes) leverages mergers and regulatory approvals, including updates to Embotrap devices, to broaden its neurointerventional offerings and support comprehensive stroke care ecosystems.
What are the Market Trends in the Blood Clot Retrieval Devices Market?
- Increasing integration of AI and machine learning for predictive clot analysis and procedural guidance.
- Shift toward hybrid devices combining aspiration and stenting for versatile clot management.
- Growing emphasis on minimally invasive techniques to reduce patient recovery times and complications.
- Rising adoption in emerging markets due to healthcare infrastructure improvements and awareness initiatives.
- Development of bioresorbable and drug-eluting devices to minimize long-term risks.
- Expansion of indications beyond stroke to include peripheral and pulmonary applications.
- Focus on cost-effective, single-use devices to address affordability in developing regions.
What Market Segments and their Subsegments are Covered in the Blood Clot Retrieval Devices Market Report?
By Device Type- Stent Retrievers
- Aspiration Devices
- Mechanical Embolus Removal Devices
- Guidewires
- Embolectomy Balloon Catheters
- Ultrasound Assisted Devices
- Thrombectomy Systems
- Catheter Directed Thrombolysis Devices
- Inferior Vena Cava Filters
- Flow Restoration Devices
- Others
- Ischemic Stroke
- Hemorrhagic Stroke
- Transient Ischemic Attack
- Acute Stroke
- Large Vessel Occlusion
- Medium Vessel Occlusion
- Distal Occlusion
- Cardioembolic Stroke
- Atherosclerotic Stroke
- Cryptogenic Stroke
- Others
- Neurovascular
- Cardiovascular
- Peripheral Vascular
- Coronary Arteries
- Cerebral Arteries
- Pulmonary Embolism
- Deep Vein Thrombosis
- Renal Vein Thrombosis
- Hepatic Vein Thrombosis
- Mesenteric Vein Thrombosis
- Others
- Hospitals
- Ambulatory Surgical Centers
- Diagnostic Centers
- Specialty Clinics
- Research Institutes
- Academic Institutions
- Emergency Medical Services
- Rehabilitation Centers
- Cardiac Centers
- Neurology Clinics
- Others
By Region
- North America
- U.S.
- Canada
- Europe
- UK
- Germany
- France
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Rest of Asia Pacific
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Middle East & Africa
- UAE
- South Africa
- Rest of Middle East & Africa
Frequently Asked Questions
The blood clot retrieval devices market involves the creation and supply of medical tools used to extract clots from blood vessels to restore normal circulation.
Key factors encompass rising stroke cases, technological progress in devices, expanding healthcare access, and supportive regulatory environments.
The market is anticipated to expand from about USD 2.3 billion in 2026 to USD 7.1 billion by 2035.
The CAGR is projected at 14.5% over the forecast period.
North America will contribute significantly, owing to sophisticated infrastructure and elevated disease rates.
Major players include Medtronic plc, Stryker Corporation, Boston Scientific Corporation, Penumbra, Inc., and Johnson & Johnson (DePuy Synthes).
The report delivers in-depth analysis covering size, trends, segments, regional overviews, key players, and projections.
Stages encompass raw material procurement, device manufacturing, clinical testing, distribution, and healthcare delivery.
Trends favor AI-enhanced and hybrid devices, with preferences shifting to minimally invasive, efficient solutions for better outcomes.
Stringent FDA approvals for safety and evolving sustainability standards in manufacturing influence innovation and market entry.