Absorbable Tissue Spacer Market Size, Share and Trends 2026 to 2035
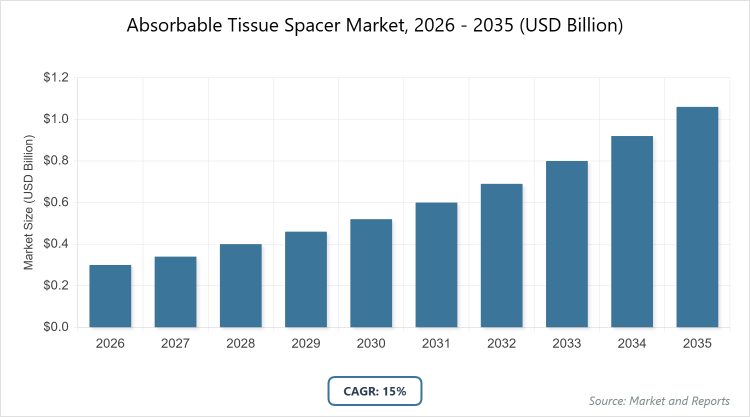
According to MarketnReports, the global Absorbable Tissue Spacer market size was estimated at USD 0.3 billion in 2025 and is expected to reach USD 1.2 billion by 2035, growing at a CAGR of 15% from 2026 to 2035. The Absorbable Tissue Spacer Market is driven by the increasing prevalence of cancer and advancements in radiation therapy techniques.
What is the Industry Overview of Absorbable Tissue Spacer?
The Absorbable Tissue Spacer market encompasses biodegradable medical devices inserted between organs or tissues to create temporary space during procedures like radiation therapy, preventing damage to adjacent healthy tissues while allowing precise targeting of diseased areas. Market definition includes hydrogel or polymer-based spacers that dissolve naturally over time, eliminating the need for removal surgeries, supporting minimally invasive treatments in oncology and other fields while addressing challenges in biocompatibility, regulatory approvals, and integration with imaging technologies for accurate placement and efficacy.
What are the Key Insights into the Absorbable Tissue Spacer?
- The global Absorbable Tissue Spacer market was valued at USD 0.3 billion in 2025 and is projected to reach USD 1.2 billion by 2035.
- The market is expected to grow at a CAGR of 15% during the forecast period from 2026 to 2035.
- The market is driven by rising cancer incidence, demand for minimally invasive therapies, technological advancements in biomaterials, and favorable reimbursement policies.
- In the type segment, hydrogel-based spacers dominate with a 60% share due to their biocompatibility and ease of injection, ideal for radiation therapy.
- In the application segment, radiation oncology dominates with a 70% share as it protects healthy tissues during radiotherapy, crucial for prostate cancer treatment.
- In the end-user segment, hospitals dominate with a 50% share owing to high-volume procedures and advanced infrastructure.
- North America dominates the regional market with a 45% share, driven by advanced healthcare systems, high cancer rates, and R&D investments in the US.
What are the Market Dynamics of Absorbable Tissue Spacer?
Growth Drivers
The Absorbable Tissue Spacer market is propelled by the increasing global burden of cancer, particularly prostate and breast cancers, where spacers minimize radiation side effects like rectal toxicity, improving patient outcomes and quality of life during treatments. Advancements in biomaterial science enable the development of safer, more effective spacers with controlled degradation rates, expanding applications in urology and gynecology. Favorable regulatory approvals from the FDA and EMA for innovative devices accelerate market entry, while growing adoption of minimally invasive procedures reduces surgical risks and hospital stays. Reimbursement policies in developed countries and rising awareness among oncologists further drive demand, supported by clinical trials demonstrating efficacy.
Restraints
High costs associated with spacer devices and procedures limit accessibility in low-income regions, where traditional radiotherapy remains preferred due to budget constraints. Limited clinical data for long-term safety and efficacy in certain applications hinders widespread adoption, requiring more extensive trials. Regulatory hurdles for new biomaterials delay approvals, increasing development expenses. Potential adverse reactions like inflammation or migration pose risks, deterring physicians. Supply chain issues for specialized hydrogels affect availability, while competition from non-absorbable alternatives offers cheaper options in some cases.
Opportunities
Opportunities arise from expanding applications in emerging therapies like brachytherapy and immunotherapy, where spacers enhance precision and reduce complications. Partnerships between biomaterial firms and medical device companies can accelerate innovation in customizable spacers. Growth in emerging markets with rising cancer rates offers potential for affordable, localized production. Integration with imaging technologies like MRI for real-time placement improves accuracy, opening niches in precision medicine. Development of multi-functional spacers with drug delivery capabilities presents futuristic avenues for combination therapies.
Challenges
Challenges include ensuring consistent spacer degradation without residues, requiring ongoing R&D to prevent complications. Variability in patient anatomy demands customized solutions, complicating mass production. Reimbursement inconsistencies across regions affect market penetration. Shortage of trained specialists for spacer placement slows adoption in underserved areas. Environmental concerns over non-biodegradable components necessitate greener materials, adding complexity. Evolving cancer treatment protocols may shift preferences away from spacers in some cases.
Absorbable Tissue Spacer Market: Report Scope
| Report Attributes | Report Details |
| Report Name | Absorbable Tissue Spacer Market |
| Market Size 2025 | USD 0.3 Billion |
| Market Forecast 2035 | USD 1.2 Billion |
| Growth Rate | CAGR of 15% |
| Report Pages | 220 |
| Key Companies Covered | Boston Scientific Corporation, Palette Life Sciences, BioProtect Ltd., Qfix, Augmenix, Inc. (Boston Scientific), Sirtex Medical Limited, Teleflex Incorporated, Varian Medical Systems, and Others |
| Segments Covered | By Type, By Application, By End-User, and By Region |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, and The Middle East and Africa (MEA) |
| Base Year | 2025 |
| Historical Year | 2020 – 2024 |
| Forecast Year | 2026 – 2035 |
| Customization Scope | Avail customized purchase options to meet your exact research needs. |
What is the Market Segmentation of Absorbable Tissue Spacer?
The Absorbable Tissue Spacer market is segmented by type, application, end-user, and region.
By Type. Hydrogel-based spacers are the most dominant subsegment, holding approximately 60% market share, due to their injectable nature and biocompatibility, facilitating easy placement. This dominance drives the market by enabling minimally invasive procedures that reduce patient discomfort and hospital costs in oncology. Hyaluronic acid-based spacers rank as the second most dominant, with around 20% share, offering natural absorption, propelling growth through applications in soft tissue separation.
By Application. Radiation oncology emerges as the most dominant subsegment, capturing about 70% share, primarily because of its role in protecting organs during radiotherapy. This leads to market growth by improving treatment safety and efficacy for cancers like prostate. Urology follows as the second most dominant, with roughly 15% share, supporting procedures like brachytherapy, driving the market via specialized medical needs.
By End-User. Hospitals represent the most dominant subsegment at about 50% share, driven by comprehensive cancer care facilities. This dominance accelerates market expansion through high-volume treatments. Ambulatory surgical centers rank second most dominant, holding around 25% share, due to outpatient procedure trends, contributing to growth via cost-effective care.
What are the Recent Developments in Absorbable Tissue Spacer?
- In January 2025, Boston Scientific expanded its SpaceOAR Vue hydrogel spacer for improved visibility in CT imaging during radiation therapy.
- In October 2024, Palette Life Sciences launched a new biodegradable spacer for gynecological applications.
- In July 2024, Augmenix (Boston Scientific) received FDA clearance for an enhanced version of its absorbable spacer.
- In April 2024, Qfix partnered with a biomaterial firm for custom tissue spacers in oncology.
- In February 2024, BioProtect raised funding for its balloon spacer in prostate cancer treatment.
What is the Regional Analysis of Absorbable Tissue Spacer?
- North America to dominate the global market.
North America holds the largest share at approximately 35%, with the United States as the dominating country, owing to high disposable incomes, wellness trends, and real estate innovation. This region’s leadership is supported by consumer demand for health-integrated homes, strong developer investments, and certifications like WELL Building Standard, fostering premium property values. Wellness communities in California and Florida lead with integrated spas and green spaces. High healthcare spending drives senior living facilities with medical wellness. Biotech hubs in Boston incorporate health-focused office designs.
Government incentives for green building boost eco-wellness developments. Venture capital funds innovative wellness tech integrations. Collaborative networks with Canada enhance cross-border projects. Focus on mental health post-pandemic increases biophilic office spaces. Luxury markets in New York prioritize wellness amenities for high-net-worth buyers. Corporate wellness programs extend to office campuses. Strong real estate financing supports large-scale community developments. Urban renewal projects in Chicago adopt wellness retrofits.
Europe follows with steady growth, driven by sustainability focus and urban wellness, where the United Kingdom dominates through luxury developments and spa integrations. The region’s expansion benefits from EU green building directives and aging population needs for senior wellness living. Nordic countries emphasize eco-wellness homes with natural materials. Germany’s engineering excellence promotes wellness-oriented offices. Multilingual compliance aids diverse markets like France and Italy. REACH regulations ensure safe material usage. Collaborative research networks advance biophilic designs. Aging infrastructure renewal projects adopt wellness retrofits. Vocational training centers build expertise in sustainable construction. Circular economy initiatives recycle building materials. Green deal policies promote energy-efficient wellness resorts. Collaborative efforts across borders enhance knowledge sharing. Rising health tourism in Spain boosts wellness hotels. Focus on work-life balance increases corporate wellness campuses.
Asia Pacific is the fastest-growing region, exhibiting high CAGR, with China leading due to urbanization and rising middle-class health awareness. This area’s potential is amplified by government support for eco-cities and wellness tourism in India and Japan. Massive urban centers in Shanghai drive demand for wellness apartments. India’s wellness retreats in Himalayas attract domestic tourism. Japan’s onsen culture integrates traditional wellness in modern developments. South Korea’s K-beauty influence boosts spa-focused properties. Cultural emphasis on holistic living accelerates adoption. Export-oriented policies enhance global competitiveness. Rising middle-class consumption increases luxury wellness homes. Environmental regulations push for green building materials. Vocational programs build expertise in wellness architecture. Collaborative R&D with global firms advances local designs. High-speed rail connectivity boosts resort developments.
Latin America demonstrates moderate progress, dominated by Mexico’s wellness resorts and tourism, supported by natural landscapes though challenged by economic variability; growth is aided by foreign investments. Brazil’s eco-communities in Amazon regions attract sustainable buyers. Government tourism initiatives in Costa Rica promote wellness retreats. The rise of middle-class health awareness in Argentina creates demand for urban wellness homes. However, economic fluctuations affect consistent investments. Emerging medical tourism in Colombia adopts wellness facilities. Regional trade agreements facilitate material imports. Vocational programs in Peru build construction skills. Biodiversity concerns influence eco-friendly designs. Urban expansion drives wellness office spaces.
The Middle East and Africa remain emerging, with the United Arab Emirates leading through luxury wellness properties and medical tourism, limited by access but promising via diversification projects. Saudi Arabia’s Vision 2030 funds wellness resorts in Red Sea areas. South Africa’s eco-lodges integrate wellness with nature tourism. Technology partnerships with European firms build expertise in Egypt. However, water scarcity impacts landscaping designs. Investments in solar-powered wellness facilities address energy needs. OPEC policies stabilize tourism-related applications. Vocational initiatives in Nigeria train for future jobs. Emerging health tourism in Morocco boosts spa developments. Focus on sustainable development goals promotes green wellness innovations. Oil-funded luxury projects in Qatar drive high-end properties. Collaborative regional efforts enhance knowledge sharing.
What are the Key Market Players in Absorbable Tissue Spacer?
- Boston Scientific Corporation. Boston Scientific focuses on hydrogel spacers like SpaceOAR, investing in imaging enhancements for precise placement.
- Palette Life Sciences. Palette develops biodegradable spacers, pursuing clinical trials for expanded applications.
- BioProtect Ltd. BioProtect specializes in balloon spacers, strategizing on funding for market expansion.
- Qfix. Qfix partners for custom spacers, focusing on oncology accessories integration.
- Augmenix, Inc. (Boston Scientific). Augmenix emphasizes absorbable technology, pursuing regulatory clearances for new indications.
- Sirtex Medical Limited. Sirtex invests in radiation therapy spacers, targeting oncology partnerships.
- Teleflex Incorporated. Teleflex develops injectable spacers, expanding in urology applications.
- Varian Medical Systems. Varian integrates spacers with radiotherapy systems, focusing on precision treatments.
What are the Market Trends in Absorbable Tissue Spacer?
- Increasing adoption of hydrogel-based spacers for biocompatibility.
- Rise of imaging-integrated spacers for accurate placement.
- Growth in minimally invasive oncology applications.
- Expansion of biodegradable polymers for reduced complications.
- Focus on cost-effective solutions for emerging markets.
- Integration with AI for treatment planning.
What Market Segments and Subsegments are Covered in the Absorbable Tissue Spacer Report?
By Type
- Hydrogel-Based Spacers
- Hyaluronic Acid-Based Spacers
- Biodegradable Polymer Spacers
- Collagen-Based Spacers
- Synthetic Absorbable Spacers
- Natural Absorbable Spacers
- Injectable Spacers
- Implantable Spacers
- Radiation Therapy Spacers
- Surgical Spacers
- Others
By Application
- Radiation Oncology
- Urology
- Gynecology
- General Surgery
- Orthopedics
- Cardiology
- Neurology
- Gastroenterology
- Plastic Surgery
- Dental Surgery
- Others
By End-User
- Hospitals
- Ambulatory Surgical Centers
- Specialty Clinics
- Cancer Treatment Centers
- Research Institutions
- Diagnostic Centers
- Academic Medical Centers
- Private Practices
- Government Healthcare Facilities
- Pharmaceutical Companies
- Others
By Region
-
- North America
- U.S.
- Canada
- Europe
- UK
- Germany
- France
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Rest of Asia Pacific
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Middle East & Africa
- UAE
- South Africa
- Rest of Middle East & Africa
- North America
Frequently Asked Questions
Absorbable Tissue Spacers are biodegradable medical devices used to separate tissues during treatments like radiation therapy to protect healthy organs.
Key factors include cancer prevalence, minimally invasive therapy demand, biomaterial advancements, and reimbursement policies.
The market is projected to grow from USD 0.3 billion in 2025 to USD 1.2 billion by 2035.
The CAGR is expected to be 15%.
North America will contribute notably, holding around 45% share due to advanced healthcare and R&D.
Major players include Boston Scientific Corporation, Palette Life Sciences, BioProtect Ltd., Qfix, and Augmenix, Inc.
The report provides detailed analysis of size, trends, segments, regional outlook, key players, and forecasts.
Stages include biomaterial sourcing, device manufacturing, clinical testing, regulatory approval, distribution, and post-market surveillance.
Trends evolve toward imaging-integrated and biodegradable spacers, with preferences for safe, effective devices.
FDA/EMA approvals ensure safety, while environmental concerns promote biodegradable materials.