RNAi for the Therapeutics Market Size, Share and Trends 2026 to 2035
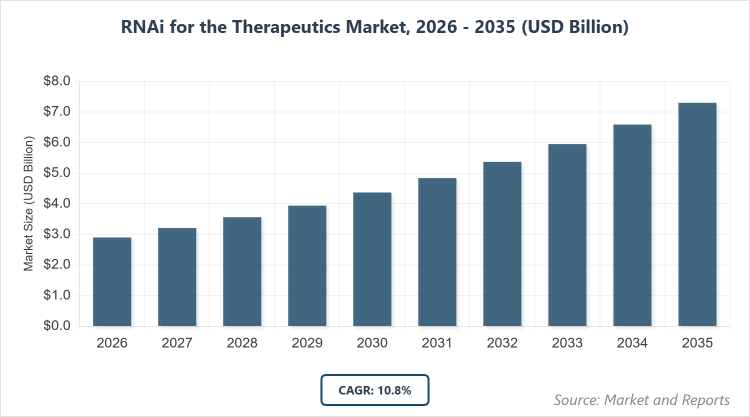
According to MarketnReports, the global RNAi for the Therapeutics market size was estimated at USD 2.9 billion in 2025 and is expected to reach USD 9.1 billion by 2035, growing at a CAGR of 10.8% from 2026 to 2035. Increasing prevalence of chronic diseases and advancements in gene silencing technologies.
What are the Key Insights into RNAi for the Therapeutics Market?
- The global RNAi for the Therapeutics market was valued at USD 2.9 billion in 2025 and is projected to reach USD 9.1 billion by 2035.
- The market is expected to grow at a CAGR of 10.8% during the forecast period from 2026 to 2035.
- The market is driven by rising demand for targeted therapies, advancements in delivery systems, and increasing approvals of RNAi-based drugs.
- The siRNA subsegment dominates the type segment with a 64% share due to its high specificity and efficacy in gene silencing, enabling treatments for rare diseases and oncology; the oncology subsegment leads the application segment with a 35% share owing to its ability to target cancer-causing genes effectively; the hospitals & clinics subsegment holds a 50% share in the end-user segment as it facilitates direct patient administration and monitoring of therapies.
- North America dominates with a 45% share due to robust R&D infrastructure, high investment in biotechnology, and favorable regulatory environment in countries like the US.
What is the RNAi for the Therapeutics Market?
Industry Overview
The RNAi for the Therapeutics market involves the development and application of RNA interference technologies to silence specific genes responsible for disease progression, offering targeted treatments for conditions such as cancer, genetic disorders, and viral infections. Market definition encompasses therapeutics utilizing small interfering RNA (siRNA), microRNA (miRNA), and short hairpin RNA (shRNA) to modulate gene expression at the post-transcriptional level, providing precision medicine alternatives to traditional drugs by addressing root causes at the molecular level and minimizing off-target effects in clinical settings.
What are the Market Dynamics in RNAi for the Therapeutics Market?
Growth Drivers
The increasing prevalence of chronic and genetic diseases, such as cancer and rare disorders, drives the RNAi therapeutics market by creating demand for innovative, gene-specific treatments that traditional therapies cannot address effectively. Advancements in delivery technologies, including lipid nanoparticles and conjugate systems, enhance the stability and targeting of RNAi molecules, enabling broader clinical applications and accelerating market adoption. Growing investments from pharmaceutical companies and government funding support extensive R&D, leading to a robust pipeline of RNAi drugs and faster regulatory approvals.
Restraints
High development and manufacturing costs for RNAi therapeutics, due to complex synthesis and specialized delivery requirements, limit accessibility and slow market penetration in resource-constrained regions. Off-target effects and immune responses pose safety concerns, requiring extensive clinical testing that delays product launches and increases overall expenses. Stringent regulatory hurdles and lengthy approval processes further constrain growth by prolonging time-to-market for new therapies.
Opportunities
Expansion into extrahepatic targets, such as the CNS and lungs, through innovative delivery platforms opens new therapeutic areas for RNAi, addressing unmet needs in neurology and respiratory diseases. Collaborations between biotech firms and big pharma accelerate pipeline development and commercialization, leveraging combined expertise and resources for faster innovation. Rising focus on personalized medicine creates opportunities for tailored RNAi therapies, enhancing efficacy for individual genetic profiles.
Challenges
Overcoming delivery barriers to non-liver tissues remains a key challenge, as current systems struggle with efficient targeting and bioavailability in diverse organs. Managing potential long-term toxicities and immune activations requires ongoing research to refine RNAi designs and minimize adverse effects. Intellectual property disputes and patent expirations could intensify competition, potentially eroding market share for established players.
RNAi for the Therapeutics Market: Report Scope
| Report Attributes | Report Details |
| Report Name | RNAi for the Therapeutics Market |
| Market Size 2025 | USD 2.9 Billion |
| Market Forecast 2035 | USD 9.1 Billion |
| Growth Rate | CAGR of 10.8% |
| Report Pages | 220 |
| Key Companies Covered |
Alnylam Pharmaceuticals, Arrowhead Pharmaceuticals, Silence Therapeutics, Arbutus Biopharma, Novo Nordisk, Sanofi, Ionis Pharmaceuticals, Sarepta Therapeutics, Benitec Biopharma, OliX Pharmaceuticals, and Others |
| Segments Covered | By Type, By Application, By End-User, and By Region |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, and The Middle East and Africa (MEA) |
| Base Year | 2025 |
| Historical Year | 2020 – 2024 |
| Forecast Year | 2026 – 2035 |
| Customization Scope | Avail customized purchase options to meet your exact research needs. |
How is the Market Segmentation of RNAi for Therapeutics?
The RNAi for the Therapeutics market is segmented by type, application, end-user, and region.
Based on Type Segment, the siRNA subsegment is the most dominant with a 64% share, due to its precise gene silencing mechanism that effectively treats genetic mutations and viral infections, driving market growth by enabling breakthrough approvals and expanding clinical applications; the miRNA subsegment is the second most dominant, offering multi-gene regulation capabilities that support complex disease management and contribute to innovative therapeutic pipelines.
Based on Application Segment, the oncology subsegment is the most dominant with a 35% share, as it targets cancer-specific genes to inhibit tumor growth and metastasis, propelling market expansion through successful clinical outcomes and increased adoption in precision oncology; the genetic disorders subsegment is the second most dominant, addressing inherited conditions at the molecular level to provide long-term symptom relief and fuel growth via orphan drug designations.
Based on End-User Segment, the hospitals & clinics subsegment is the most dominant with a 50% share, facilitating direct patient care and administration of RNAi therapies, which drives market growth by integrating advanced treatments into standard healthcare practices; the research institutes subsegment is the second most dominant, conducting pivotal studies that validate efficacy and safety, accelerating innovation and regulatory progress.
What are the Recent Developments in RNAi for the Therapeutics Market?
- In February 2026, SanegeneBio announced a global licensing collaboration with Genentech to advance an RNAi therapeutic program, focusing on novel targets for undisclosed diseases.
- In January 2026, Alnylam Pharmaceuticals launched its “Alnylam 2030” strategy, aiming to expand ATTR amyloidosis leadership and drive growth through innovative RNAi pipelines.
- In September 2025, Novartis entered a USD 4.165 billion collaboration with Shanghai Argo to develop cardiovascular RNAi assets, enhancing its portfolio in gene silencing therapies.
- In March 2025, Alnylam Pharmaceuticals received FDA approval for Qfitlia (fitusiran), an RNAi therapeutic for hemophilia A and B, marking the sixth approved RNAi drug.
How is the Regional Analysis of RNAi for the Therapeutics Market?
- North America is expected to dominate the global market.
North America leads the RNAi for the Therapeutics market with a 45% share, driven by advanced healthcare infrastructure and significant R&D investments in the United States, the dominant country, where numerous FDA approvals and biotech hubs accelerate innovation and commercialization.
Europe holds a 25% share, supported by strong regulatory frameworks and collaborations in Germany, the leading country, focusing on oncology and rare diseases to advance clinical trials and market access.
Asia-Pacific accounts for a 20% share, with rapid growth fueled by increasing biotechnology investments in China, the key country, emphasizing affordable RNAi solutions for infectious diseases and expanding manufacturing capabilities.
Latin America represents a 5% share, led by Brazil as the dominant country, where government initiatives promote research in genetic disorders and partnerships enhance access to emerging therapies.
The Middle East and Africa hold a 5% share, with the UAE emerging as a hub through investments in precision medicine and collaborations addressing regional health challenges like metabolic diseases.
Who are the Key Market Players in RNAi for the Therapeutics?
- Alnylam Pharmaceuticals pioneers RNAi therapeutics with approved drugs like Onpattro and Amvuttra, focusing on long-term service agreements and digital innovations to expand access in rare diseases.
- Arrowhead Pharmaceuticals develops targeted RNAi molecules using its TRiM platform, emphasizing partnerships with pharma giants to advance clinical pipelines in liver and pulmonary diseases.
- Silence Therapeutics specializes in siRNA stabilization technology, pursuing strategic alliances for oncology and cardiovascular programs to accelerate development and commercialization.
- Arbutus Biopharma advances lipid nanoparticle delivery systems for RNAi, collaborating on hepatitis B therapies to enhance efficacy and reduce dosing frequency.
- Novo Nordisk integrates RNAi into its cardiometabolic portfolio through acquisitions and collaborations, targeting obesity and diabetes with gene-silencing approaches.
- Sanofi leverages RNAi for immunology and rare diseases, investing in platform technologies to build a diverse pipeline via external partnerships.
- Ionis Pharmaceuticals focuses on antisense RNA but incorporates RNAi elements, emphasizing neurological disorders through co-development agreements.
- Sarepta Therapeutics combines RNAi with exon-skipping for muscular dystrophy, prioritizing pediatric approvals and global expansion strategies.
- Benitec Biopharma employs DNA-directed RNAi for ocular and infectious diseases, pursuing orphan designations to streamline regulatory paths.
- OliX Pharmaceuticals develops asymmetric siRNA for dermatology and ophthalmology, focusing on topical delivery innovations and Asian market penetration.
What are the Market Trends in RNAi for Therapeutics?
- Expansion of extrahepatic delivery systems to target the CNS, lungs, and muscles, broadening therapeutic applications beyond liver diseases.
- Increasing focus on combination therapies integrating RNAi with immunotherapies for enhanced efficacy in oncology.
- Growth in personalized medicine through biomarker-driven RNAi designs for tailored patient treatments.
- Rising adoption of long-acting formulations to reduce dosing frequency and improve patient compliance.
- Surge in strategic partnerships between biotech firms and big pharma for accelerated clinical development.
What Market Segments and Subsegments are Covered in the RNAi for the Therapeutics Report?
By Type
- siRNA
- miRNA
- shRNA
By Application
- Oncology
- Genetic Disorders
- Infectious Diseases
- Neurological Disorders
- Cardiovascular Diseases
By End-User
- Hospitals & Clinics
- Research Institutes
- Pharmaceutical Companies
By Region
-
- North America
- U.S.
- Canada
- Europe
- UK
- Germany
- France
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Rest of Asia Pacific
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Middle East & Africa
- UAE
- South Africa
- Rest of Middle East & Africa
- North America
Frequently Asked Questions
RNAi for therapeutics involves using RNA interference to silence specific genes, providing targeted treatments for diseases like cancer and genetic disorders by modulating gene expression.
Key factors include advancements in delivery technologies, rising chronic disease prevalence, increasing regulatory approvals, and growing investments in precision medicine.
The market is projected to grow from USD 3.6 billion in 2026 to USD 9.1 billion by 2035.
The CAGR is expected to be 10.8% from 2026 to 2035.
North America will contribute notably, holding around 45% of the market share due to strong R&D and regulatory support.
Major players include Alnylam Pharmaceuticals, Arrowhead Pharmaceuticals, Silence Therapeutics, Arbutus Biopharma, and Novo Nordisk.
The report offers in-depth analysis of market size, trends, segmentation, regional insights, key players, and forecasts from 2026 to 2035.
Stages include target identification, molecule design, preclinical testing, clinical trials, manufacturing, regulatory approval, and commercialization.
Trends are shifting toward extrahepatic targeting and personalized therapies, with preferences for minimally invasive, long-acting treatments that offer high efficacy and fewer side effects.
Favorable FDA approvals for orphan drugs and fast-track designations accelerate growth, while environmental concerns over nanoparticle delivery materials influence sustainable development practices.