Fosfomycin Trometamol Market Size and Forecast 2026 to 2035
According to MarketnReports, the global Fosfomycin Trometamol market size was estimated at USD 39.77 Million in 2025 and is expected to reach USD 54.78 Million by 2035, growing at a CAGR of 3.4% from 2026 to 2035. Fosfomycin Trometamol Market is driven by the rising global incidence of urinary tract infections (UTIs) and the increasing need for effective treatments against multidrug-resistant bacteria.
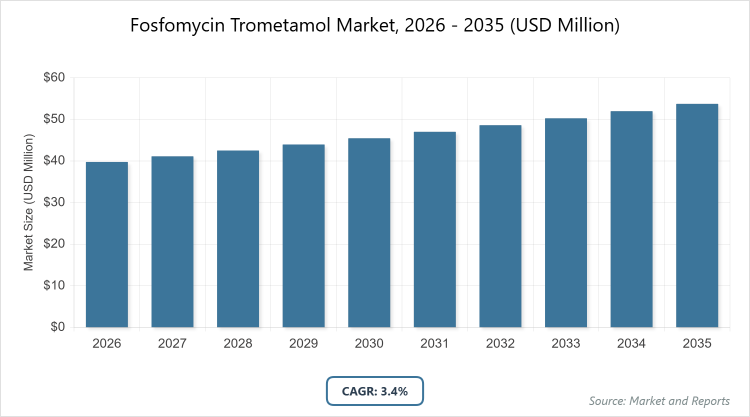
What are the Key Insights into the Fosfomycin Trometamol Market?
- The global Fosfomycin Trometamol market is projected to grow at a CAGR of 3.4% from 2026 to 2035.
- The market value is expected to increase from approximately USD 39.77 Million in 2025 to USD 54.78 Million by 2035.
- In the Application segment, the Postoperative Urinary Tract Infection subsegment is expected to dominate.
- In the Type segment, the Fosfomycin Tromethamine Powder subsegment is anticipated to lead.
- North America is projected to be the dominant region.
What is the Fosfomycin Trometamol Industry Overview?
The Fosfomycin Trometamol market revolves around a specialized antibiotic primarily used for treating uncomplicated urinary tract infections (UTIs), particularly acute cystitis in women. It functions by inhibiting bacterial cell wall synthesis, effectively targeting both gram-positive and gram-negative bacteria responsible for bladder infections. Available in powder or granule form, this medication is odorless, white crystalline in appearance, and soluble in water or methanol but not in chloroform.
It is prescribed for oral administration as a single dose, typically mixed with water, and is valued for its broad-spectrum antibacterial properties. The market encompasses the production, distribution, and consumption of this drug, driven by the need for effective treatments against common lower urinary tract issues, while facing scrutiny due to potential side effects and the necessity for medical supervision to avoid misuse that could lead to antibiotic resistance or adverse reactions.
What are the Market Dynamics in the Fosfomycin Trometamol Market?
Growth Drivers
The Fosfomycin Trometamol market is propelled by the escalating incidence of urinary tract infections, especially among women, where factors such as female anatomy, sexual activity, menopause, and certain birth control methods heighten vulnerability. This antibiotic’s efficacy in treating lower tract infections like cystitis, combined with global statistics indicating that up to 50% of women experience UTIs at least once in their lifetime, fuels demand. Additionally, investments in advanced urology centers equipped with modern diagnostics and treatments enhance accessibility, enabling timely intervention and boosting market expansion through improved healthcare infrastructure.
Restraints
Market growth is hindered by the medication’s associated side effects, including nausea, diarrhea, headache, dizziness, and vaginal discomfort, which may deter usage or require additional medical attention. Rare but severe risks, such as intestinal conditions triggered by C. difficile bacteria, add to concerns, potentially limiting prescriptions. Furthermore, the drug’s restricted application to uncomplicated bladder infections excludes it from treating more severe or upper tract UTIs, narrowing its market scope and encouraging reliance on alternative antibiotics.
Opportunities
Opportunities arise from expanding regulatory approvals for generic versions in new markets, exemplified by launches like Xiromed LLC’s Fosfomycin Tromethamine Granules, which broaden availability and affordability. Ongoing research into UTI-causing bacteria, such as studies using advanced microscopy to understand pathogen behavior, paves the way for innovative detection and proactive treatments, potentially integrating Fosfomycin Trometamol into advanced therapeutic protocols and opening avenues for market penetration in emerging regions with rising healthcare awareness.
Challenges
The market faces challenges from alternative UTI treatments like cephalexin, nitrofurantoin, or ceftriaxone, which offer broader applicability for infections beyond the bladder. Physician preferences and subjective treatment approaches vary, impacting adoption, while the drug’s single-dose requirement and potential for incomplete efficacy in complex cases pose barriers. Additionally, global variations in healthcare access and awareness complicate uniform market growth.
Table Code (Report Scope)
Fosfomycin Trometamol Market: Report Scope
| Report Attributes | Report Details |
| Report Name | Fosfomycin Trometamol Market |
| Market Size 2025 | USD 39.77 Million |
| Market Forecast 2035 | USD 54.78 Million |
| Growth Rate | CAGR of 3.4% |
| Report Pages | 225 |
| Key Companies Covered |
Meiji Seika Pharma, Zambon Group, CSPC Pharmaceutical Group, Sandoz, Merck & Co., S.A.L.A.R.S. Pharmaceuticals, Pfizer, Sanofi, Teva Pharmaceutical Industries, Bristol Myers Squibb, Sun Pharmaceutical Industries, North China Pharmaceutical Corporation (NCPC), Mylan, GlaxoSmithKline, Bayer, and others. |
| Segments Covered | By Type, By Application, By Region. |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, The Middle East and Africa (MEA) |
| Base Year | 2025 |
| Historical Year | 2020 – 2024 |
| Forecast Year | 2026 – 2035 |
| Customization Scope | Avail customized purchase options to meet your exact research needs. |
What is the Market Segmentation in the Fosfomycin Trometamol Market?
The Fosfomycin Trometamol market is segmented by type, application, and region.
Type Segmentation
The Fosfomycin Tromethamine Powder segment is the most dominant in the Type category, owing to its versatility in formulation, ease of manufacturing, and growing approvals for powder-based generics that enhance solubility and patient compliance; this leadership position is bolstered by its rapid absorption and effectiveness in single-dose regimens, which streamline treatment and reduce dosing errors, propelling market growth through cost-effective production and wider distribution, whereas the Fosfomycin Tromethamine Granules segment is the second most dominant, favored for its convenient oral dissolution but limited by higher historical revenue in specific markets, contributing to market expansion by offering user-friendly options that cater to diverse consumer preferences and improve adherence in outpatient settings.
Application Segmentation
The Postoperative Urinary Tract Infection segment emerges as the most dominant in the Application category, driven by its critical role in preventing and treating infections following surgical procedures, where patients are at heightened risk due to catheterization or invasive interventions; this dominance stems from the drug’s single-dose efficacy in reducing postoperative complications, thereby lowering hospital readmission rates and healthcare costs, while the Common Urinary Tract Infection segment ranks second, benefiting from the high prevalence of uncomplicated UTIs in everyday scenarios, yet it trails due to greater competition from alternative antibiotics for non-surgical cases, ultimately helping drive the overall market by addressing a broad patient base and supporting preventive healthcare strategies.
What are the Recent Developments in the Fosfomycin Trometamol Market?
- In October 2023, Brigham and Women’s Hospital established a new Department of Urology, marking a significant expansion in specialized care facilities that could increase access to Fosfomycin Trometamol for UTI treatments through advanced diagnostics and integrated therapies.
- In October 2020, Xiromed LLC received approvals to launch Fosfomycin Tromethamine Granules for Oral Solution, introducing the first AA-rated generic equivalent to Monurol®, thereby enhancing market competition and affordability for generic options in UTI management.
- In August 2022, researchers at the University of Technology Sydney utilized state-of-the-art microscopy to study the spread of Uropathogenic Escherichia coli, providing insights that could lead to earlier UTI detection and more targeted use of antibiotics like Fosfomycin Trometamol.
- In December 2021, MedicaMetrix Inc and Emerson Hospital’s Yeatts Urology Center formed a strategic partnership to create an international urology center of excellence, potentially boosting the adoption of Fosfomycin Trometamol through collaborative research and improved clinical practices.
What is the Regional Analysis of the Fosfomycin Trometamol Market?
North America to dominate the market
North America dominates the Fosfomycin Trometamol market, primarily driven by advanced healthcare infrastructure, high UTI prevalence among women, and robust FDA approvals for generics, with the United States as the leading country due to its extensive access to primary care, rising healthcare expenditures, and strong awareness campaigns that facilitate early treatment and market penetration.
Europe exhibits steady growth in the Fosfomycin Trometamol market, fueled by an aging female population, increasing UTI incidences, and supportive awareness programs, with Germany as the dominating country owing to its sophisticated pharmaceutical industry, high per capita healthcare spending, and efficient regulatory frameworks that promote antibiotic innovation and distribution.
Asia Pacific is experiencing rapid expansion in the Fosfomycin Trometamol market, attributed to improving healthcare access, growing patient awareness, and rising antibiotic resistance concerns, with China leading the region through its massive population, increasing investments in urology facilities, and domestic production capabilities that lower costs and enhance availability.
Latin America shows emerging potential in the Fosfomycin Trometamol market, supported by urbanization and better medical infrastructure, though growth is moderated by economic variations, with Brazil as the dominant country due to its large population, government initiatives for women’s health, and expanding pharmaceutical manufacturing that aids in affordable drug supply.
The Middle East & Africa region is gradually advancing in the Fosfomycin Trometamol market, driven by healthcare reforms and increasing focus on infectious disease management, with South Africa leading through its relatively advanced medical systems, awareness efforts on UTIs, and strategic imports that support treatment accessibility in underserved areas.
Who are the Key Market Players and What are Their Strategies in the Fosfomycin Trometamol Market?
- Meiji Seika Pharma focuses on research and development in antibiotics, leveraging its expertise in infectious diseases to innovate formulations and expand into Asian markets through partnerships and clinical trials.
- Zambon Group emphasizes global expansion via acquisitions and collaborations, prioritizing respiratory and urological therapies to enhance product portfolios and penetrate emerging markets with cost-effective generics.
- CSPC Pharmaceutical Group adopts a strategy of large-scale manufacturing and vertical integration, aiming to dominate in China and export markets by investing in quality control and regulatory compliance for affordable drug supply.
- Sandoz pursues generic drug leadership through Novartis-backed R&D, focusing on bioequivalence studies and market access strategies to offer low-cost alternatives and capture share in regulated regions.
- Merck & Co. invests heavily in oncology and infectious disease innovation, using mergers like with Acceleron Pharma to strengthen its antibiotic pipeline and employ data-driven portfolio management for sustained growth.
- A.L.A.R.S. Pharmaceuticals concentrates on niche antibiotic markets, employing supply chain optimization and efficiency technologies to maintain competitive pricing and expand distribution networks.
- Pfizer utilizes acquisitions such as Seagen to bolster its therapeutic areas, applying intellectual property protection and global marketing to drive antibiotic sales amid rising resistance concerns.
- Sanofi focuses on diversified portfolios through joint ventures and R&D in vaccines and antimicrobials, optimizing supply chains for cost leadership and addressing UTI treatments in diverse geographies.
- Teva Pharmaceutical Industries prioritizes generic development and cost-reduction strategies, leveraging economies of scale and regulatory expertise to flood markets with affordable Fosfomycin variants.
- Bristol Myers Squibb integrates mature product portfolios via partnerships, like with MBO for sales forces, to enhance market penetration and focus on oncology-adjacent infectious disease solutions.
- Sun Pharmaceutical Industries employs aggressive expansion in generics, investing in Indian manufacturing hubs to export low-cost drugs and form alliances for broader international reach.
- North China Pharmaceutical Corporation (NCPC) targets domestic dominance through state-supported production, emphasizing quality upgrades and exports to compete in Asia Pacific with volume-based strategies.
- Mylan (now part of Viatris) focuses on mergers for scale, streamlining operations to provide generic antibiotics and navigate regulatory landscapes for global market entry.
- GlaxoSmithKline implements purchasing optimizations and tender negotiations, investing in R&D for next-generation antimicrobials while maintaining strong sales forces for antibiotic promotion.
- Bayer leverages its pharmaceutical heritage in innovation, pursuing collaborations and patent protections to advance UTI treatments and expand in high-growth regions like Europe and Asia.
What are the Market Trends in the Fosfomycin Trometamol Market?
- Increasing prevalence of urinary tract infections (UTIs) globally, particularly among women, driving demand for effective single-dose antibiotics.
- Growing concerns over antibiotic resistance, positioning Fosfomycin Trometamol as a viable alternative due to its broad-spectrum efficacy.
- Rising adoption of generic versions, fueled by regulatory approvals and cost pressures in healthcare systems.
- Expansion of urology centers and healthcare infrastructure, enhancing accessibility and early treatment options.
- Advancements in research on pathogen behavior, leading to more targeted and proactive UTI management strategies.
- Shift toward single-dose oral administrations for patient convenience and compliance in outpatient settings.
What Market Segments are Covered in the Fosfomycin Trometamol Report?
By Type
- Fosfomycin Tromethamine Powder
- Fosfomycin Tromethamine Granules
By Application
- Postoperative Urinary Tract Infection
- Common Urinary Tract Infection
- Preoperative Urinary Tract Infection
- Others
By Region
-
- North America
- U.S.
- Canada
- Europe
- UK
- Germany
- France
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Rest of Asia Pacific
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Middle East & Africa
- UAE
- South Africa
- Rest of Middle East & Africa
- North America
Frequently Asked Questions
Fosfomycin Trometamol is an antibiotic medication used primarily for treating uncomplicated bladder infections, such as lower urinary tract infections (UTIs) and acute cystitis in women, by inhibiting bacterial cell wall synthesis.
Key factors include rising UTI prevalence, antibiotic resistance concerns, investments in urology infrastructure, generic approvals, and research innovations in pathogen detection and treatment.
The market is projected to grow from approximately USD 39.77 Million in 2025 to USD 54.78 Million by 2035.
The CAGR is expected to be 3.4% during 2026-2035.
North America will contribute notably, driven by advanced healthcare and high UTI awareness.
Major players include Meiji Seika Pharma, Zambon Group, CSPC Pharmaceutical Group, Sandoz, Merck & Co., S.A.L.A.R.S. Pharmaceuticals, Pfizer, Sanofi, Teva Pharmaceutical Industries, Bristol Myers Squibb, Sun Pharmaceutical Industries, North China Pharmaceutical Corporation (NCPC), Mylan, GlaxoSmithKline, Bayer, and others.
The report provides insights into market size, growth drivers, restraints, segmentation, regional analysis, key players, trends, and future opportunities.
The value chain includes raw material sourcing, manufacturing, quality testing, distribution, regulatory approvals, and end-user prescription and consumption.
Trends are shifting toward single-dose generics for convenience, with consumers preferring affordable, effective treatments amid growing awareness of antibiotic stewardship.
Regulatory approvals for generics boost growth, while environmental concerns over pharmaceutical waste and strict antibiotic use guidelines to combat resistance pose challenges.