E-Clinical Solution Software Market Size, Share and Trends 2026 to 2035
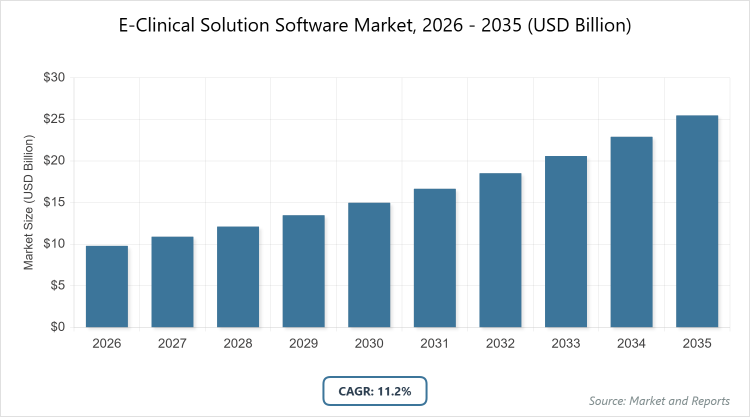
According to MarketnReports, the global E-Clinical Solution Software market size was estimated at USD 9.8 billion in 2025 and is expected to reach USD 28.5 billion by 2035, growing at a CAGR of 11.2% from 2026 to 2035. The E-Clinical Solution Software Market is driven by increasing clinical trials, regulatory compliance needs, and the adoption of digital tools for efficient data management in pharmaceutical research.
What are the Key Insights into E-Clinical Solution Software?
- The global E-Clinical Solution Software market was valued at USD 9.8 billion in 2025 and is projected to reach USD 28.5 billion by 2035.
- The market is expected to grow at a CAGR of 11.2% during the forecast period from 2026 to 2035.
- The market is driven by rising clinical trial complexity, digital transformation in pharma, AI integration for data analysis, and stringent regulatory requirements.
- In the type segment, Electronic Data Capture (EDC) Software dominates with a 35% share due to its role in efficient data collection and real-time monitoring.
- In the application segment, Clinical Trials dominates with a 40% share as it streamlines trial management and compliance.
- In the end-user segment, Pharmaceutical Companies dominate with a 45% share owing to high R&D investments and need for integrated platforms.
- North America dominates the regional market with a 40% share, driven by advanced pharma R&D, regulatory support, and presence of key players like Oracle and Medidata.
What is the Industry Overview of E-Clinical Solution Software?
The E-Clinical Solution Software market comprises digital platforms and tools that streamline clinical trial processes, including data capture, management, analysis, and reporting, enhancing efficiency, accuracy, and compliance in drug development and medical research. Market definition includes cloud-based and on-premise systems like EDC, CTMS, and eTMF that facilitate real-time collaboration, reduce paperwork, and ensure data integrity while addressing challenges in interoperability, data security, and integration with emerging technologies like AI for predictive analytics in a highly regulated healthcare environment.
What are the Market Dynamics of E-Clinical Solution Software?
Growth Drivers
The E-Clinical Solution Software market is propelled by the surge in clinical trials driven by chronic diseases and personalized medicine, where software streamlines data collection, reduces errors, and accelerates timelines through real-time analytics and remote monitoring. Digital transformation in pharma, with AI and cloud integration, enhances collaboration across global teams, cutting costs and improving compliance with FDA/EMA standards. Increasing outsourcing to CROs demands scalable platforms for multi-site trials, while blockchain for data security addresses integrity concerns. Government initiatives for electronic health records and telehealth further drive adoption, supported by investments in big data for predictive insights.
Restraints
High implementation costs for integrated e-clinical suites and customization limit adoption among small biotechs and in emerging markets, where legacy systems persist. Data privacy concerns under GDPR/HIPAA require robust security, increasing compliance burdens and delaying deployments. Interoperability issues between platforms from different vendors hinder seamless data flow, causing inefficiencies. Shortage of skilled IT personnel for software management slows rollout in healthcare settings. Economic uncertainties reduce R&D budgets, impacting software investments.
Opportunities
Opportunities emerge from AI-driven predictive analytics for trial optimization and patient recruitment, attracting pharma giants for faster drug development. Expansion into decentralized trials with remote capabilities opens niches in telehealth and patient-centric models. Partnerships between software providers and CROs can create end-to-end solutions. Emerging markets with growing pharma sectors offer potential for cloud-based, affordable platforms. The development of blockchain for secure, tamper-proof data enhances trust in multi-center studies.
Challenges
Challenges include ensuring data security against cyber threats in cloud environments, requiring constant updates and encryption. Rapid regulatory changes demand flexible software adaptations, straining development. Integration with diverse EHR systems complicates workflows. User resistance to new technologies necessitates training, while high trial failure rates affect ROI perceptions. Global disparities in digital infrastructure hinder uniform adoption.
E-Clinical Solution Software Market: Report Scope
| Report Attributes | Report Details |
| Report Name | E-Clinical Solution Software Market |
| Market Size 2025 | USD 9.8 Billion |
| Market Forecast 2035 | USD 28.5 Billion |
| Growth Rate | CAGR of 11.2% |
| Report Pages | 220 |
| Key Companies Covered | Oracle Corporation, Medidata Solutions (Dassault Systèmes), Veeva Systems, IQVIA, Parexel International, BioClinica (ERT), eClinical Solutions, Anju Software, and Others |
| Segments Covered | By Type, By Application, By End-User, and By Region |
| Regions Covered | North America, Europe, Asia Pacific (APAC), Latin America, and The Middle East and Africa (MEA) |
| Base Year | 2025 |
| Historical Year | 2020 – 2024 |
| Forecast Year | 2026 – 2035 |
| Customization Scope | Avail customized purchase options to meet your exact research needs. |
What is the Market Segmentation of E-Clinical Solution Software?
The E-Clinical Solution Software market is segmented by type, application, end-user, and region.
By Type. Electronic Data Capture (EDC) Software is the most dominant subsegment, holding approximately 35% market share, due to its core role in accurate, real-time data collection. This dominance drives the market by reducing manual errors and speeding up trial phases in pharma R&D. Clinical Trial Management Systems (CTMS) rank as the second most dominant, with around 25% share, offering comprehensive oversight, propelling growth through efficient site management and compliance.
By Application. Clinical Trials emerge as the most dominant subsegment, capturing about 40% share, primarily because of the need for streamlined processes in drug development. This leads to market growth by facilitating faster approvals and cost savings. Drug Development follows as the second most dominant, with roughly 20% share, supporting R&D pipelines, driving the market via innovation in biotech.
By End-User. Pharmaceutical Companies represent the most dominant subsegment at about 45% share, driven by extensive trial portfolios. This dominance accelerates market expansion through large-scale implementations. Contract Research Organizations (CROs) rank second most dominant, holding around 25% share, due to outsourcing trends, contributing to growth via specialized services.
What are the Recent Developments in E-Clinical Solution Software?
- In January 2025, Medidata launched an AI-enhanced EDC platform for predictive trial insights.
- In October 2024, Oracle Health acquired a CTMS startup to integrate with its cloud suite.
- In July 2024, Veeva Systems expanded its eTMF with blockchain for secure data sharing.
- In April 2024, IQVIA partnered with an AI firm for advanced analytics in RTSM.
- In February 2024, Parexel introduced a mobile ePRO app for patient-centric trials.
What is the Regional Analysis of E-Clinical Solution Software?
- North America is expected to dominate the global market.
North America holds the largest share at approximately 40%, with the United States as the dominating country, owing to advanced pharma R&D, high trial volumes, and the presence of leaders like Medidata and Oracle. This region’s leadership is supported by FDA support for digital tools, strong VC funding for health tech, and widespread adoption in biotech hubs, fostering innovation and compliance. Major pharma clusters in Boston and San Francisco drive EDC and CTMS adoption. NIH grants fund e-clinical platform trials. Defense medical research requires secure data management. University-industry partnerships in Johns Hopkins advance analytics integration. High insurance coverage for clinical research supports facility investments. Strict FDA guidelines ensure market preference for validated systems. Reshoring of clinical operations boosts domestic demand.
Europe follows with steady growth, driven by EMA regulations and research focus, where Germany dominates through its pharma industry. The region’s expansion benefits from EU funding for digital health and collaborative trials across borders. Horizon Europe programs finance multi-center e-clinical projects. The UK’s advanced biopharma hubs in London promote adoption in cell therapy trials. Multilingual compliance aids diverse markets like France and Italy. GDPR regulations ensure secure data handling. Industry consortia share validation best practices. Aging population research adopts ePRO for patient-reported outcomes. Vocational training centers build expertise in software operations. Collaborative R&D across borders advances CTMS designs. Focus on biologic drugs post-pandemic boosts related applications. Strict export standards enhance global competitiveness.
Asia Pacific is the fastest-growing region, exhibiting a high CAGR, with India leading due to CRO growth and cost-effective trials. This area’s potential is amplified by government digital health initiatives and pharma exports in China and Japan. Massive clinical trial centers in Hyderabad drive demand for integrated EDC platforms. China’s biotech boom requires advanced CTMS for global compliance. Japan’s precision medicine tradition favors high-resolution data management. South Korea’s display industry requires regulatory software for medical devices. Cultural emphasis on quality control accelerates training programs. Export-oriented policies enhance global competitiveness. Rising middle-class consumption increases medical research demand. Environmental regulations push for cloud efficiency. Vocational programs build expertise in e-clinical applications.
Latin America demonstrates moderate progress, dominated by Brazil’s clinical research sector, supported by foreign investments, though limited by regulations; growth is aided by trial outsourcing. Mexico benefits from NAFTA ties, facilitating tech transfers from North America. Government health initiatives in Argentina promote digital platforms. The rise of generics in Chile creates demand for cost-effective software. However, currency fluctuations impact import costs for high-end platforms. Emerging CROs in Colombia adopt for global trials. Regional trade pacts like Mercosur ease software licenses. Vocational training expands in Peru for clinical applications. Biodiversity concerns influence eco-friendly data practices. Urban expansion drives research center demand.
The Middle East and Africa remain emerging, with the United Arab Emirates leading through health tech investments and medical tourism, constrained by infrastructure but promising via diversification. Saudi Arabia’s Vision 2030 funds advanced e-clinical facilities. South Africa’s universities collaborate on clinical research. Technology partnerships with European firms build expertise in Egypt. However, connectivity issues impact cloud adoption. Investments in digital health create demand for mobile ePRO. OPEC policies stabilize health-related applications. Vocational initiatives in Nigeria train for future jobs. Emerging labs in Kenya require software for medical research.
What are the Key Market Players in E-Clinical Solution Software?
- Oracle Corporation. Oracle focuses on cloud-based suites like Clinical One, investing in AI for data analytics.
- Medidata Solutions (Dassault Systèmes). Medidata emphasizes unified platforms, pursuing acquisitions for expanded capabilities.
- Veeva Systems. Veeva develops Vault eTMF, strategizing on life sciences compliance.
- IQVIA. IQVIA offers end-to-end solutions, focusing on big data integration.
- Parexel International. Parexel invests in ePRO, targeting patient-centric trials.
- BioClinica (ERT). BioClinica specializes in imaging and EDC, expanding in oncology.
- eClinical Solutions. eClinical provides Elluminate, emphasizing analytics for CROs.
- Anju Software. Anju focuses on modular tools, pursuing global partnerships.
What are the Market Trends in E-Clinical Solution Software?
- Increasing AI for predictive analytics and automation.
- Rise of decentralized and virtual trials.
- Adoption of cloud-based platforms for scalability.
- Growth in blockchain for data security.
- Expansion of ePRO for patient engagement.
- Focus on interoperability with EHR systems.
What Market Segments and Subsegments are Covered in the E-Clinical Solution Software Report?
By Type
- Electronic Data Capture (EDC) Software
- Clinical Trial Management Systems (CTMS)
- Randomization and Trial Supply Management (RTSM)
- Electronic Clinical Outcome Assessment (eCOA)
- Clinical Data Management Systems (CDMS)
- Electronic Trial Master File (eTMF)
- Safety Solutions
- Regulatory Information Management Systems (RIMS)
- Electronic Patient-Reported Outcomes (ePRO)
- Interactive Response Technology (IRT)
- Others
By Application
- Clinical Trials
- Drug Development
- Patient Data Management
- Regulatory Compliance
- Safety Monitoring
- Biostatistics
- Site Management
- Protocol Management
- Budgeting & Forecasting
- Electronic Consent
- Others
By End-User
- Pharmaceutical Companies
- Contract Research Organizations (CROs)
- Biotechnology Companies
- Academic Research Institutions
- Hospitals & Clinics
- Medical Device Manufacturers
- Government & Regulatory Bodies
- Consulting Firms
- Diagnostic Centers
- Healthcare Providers
- Others
By Region
-
- North America
- U.S.
- Canada
- Europe
- UK
- Germany
- France
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Rest of Asia Pacific
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Middle East & Africa
- UAE
- South Africa
- Rest of Middle East & Africa
- North America
Frequently Asked Questions
E-Clinical Solution Software are digital platforms for managing clinical trials, data capture, and regulatory compliance in pharmaceutical research.
Key factors include clinical trial growth, digital transformation, AI adoption, and regulatory demands.
The market is projected to grow from USD 9.8 billion in 2025 to USD 28.5 billion by 2035.
The CAGR is expected to be 11.2%.
North America will contribute notably, holding around 40% share due to R&D and regulations.
Major players include Oracle Corporation, Medidata Solutions, Veeva Systems, IQVIA, and Parexel International.
The report provides comprehensive analysis including size, trends, segments, regional outlook, key players, and forecasts.
Stages include software development, integration, deployment, training, and support services.
Trends evolve toward AI and cloud platforms, with preferences for user-friendly, compliant tools.
FDA/EMA regulations mandate data integrity, while cloud energy concerns influence sustainable hosting.